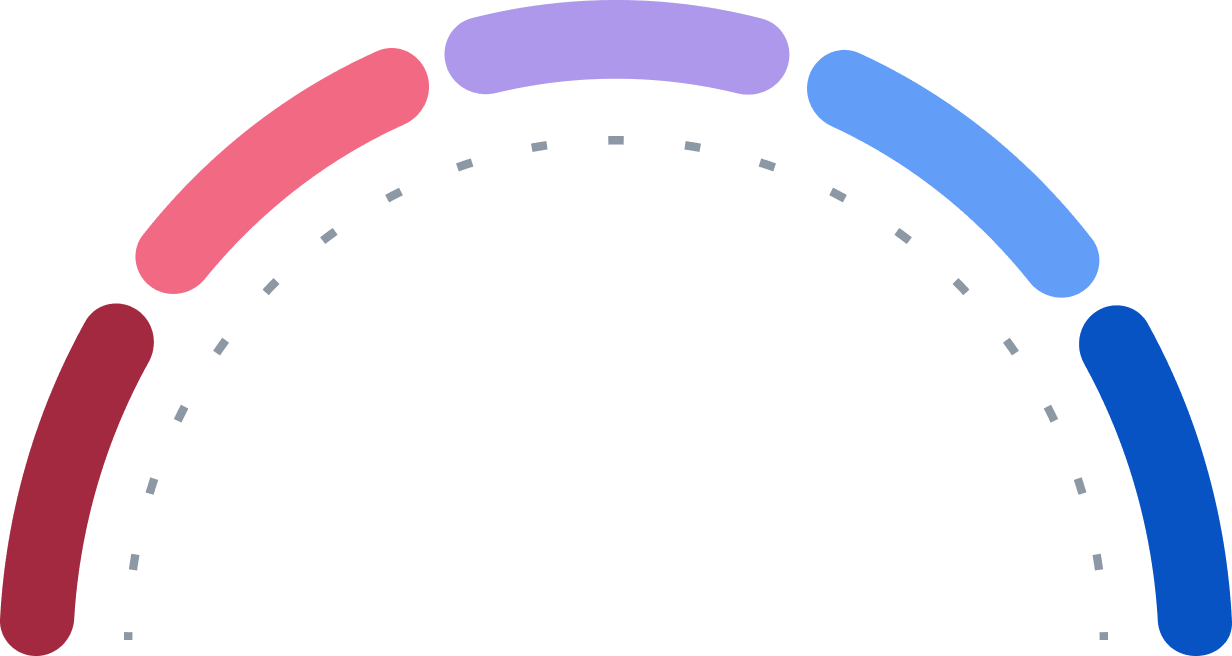
Buy
Strong Buy
Buy
Hold
Sell
Strong Sell
0
14
5
0
0


| Exchange | Ticker | Currency | Last Trade | Price | Daily Change |
|---|---|---|---|---|---|
NASDAQ |
VSTM
|
USD
|
29.04.2026 11:50
|
5,50 USD
| 0,04 USD
+0,73 %
|
Hamburg |
VIRSN35.HAMB
|
EUR
|
29.04.2026 06:11
|
4,82 EUR
| -0,30 EUR
-5,94 %
|
Quotrix |
VIRSN35.DUSD
|
EUR
|
29.04.2026 05:27
|
4,79 EUR
| -0,33 EUR
-6,42 %
|
IEX |
VSTM
|
USD
|
28.04.2026 19:59
|
5,45 USD
| -0,09 USD
-1,71 %
|
Düsseldorf |
VIRSN35.DUSB
|
EUR
|
28.04.2026 17:30
|
4,57 EUR
| -0,55 EUR
-10,80 %
|
London |
0LOV.L
|
USD
|
28.04.2026 16:04
|
5,39 USD
| -0,15 USD
-2,63 %
|
 Frankfurt
Frankfurt
| Ex-Date | Dividend per Share |
|---|---|
| 15.03.2022 | 0,02 USD |
| Date | Split |
|---|---|
| 01.06.2023 | 1:12 |
| Name | Symbol |
|---|---|
| Düsseldorf | VIRSN35.DUSB |
| Frankfurt | 2VSA.F |
| Hamburg | VIRSN35.HAMB |
| London | 0LOV.L |
| NASDAQ | VSTM |
| Quotrix | VIRSN35.DUSD |