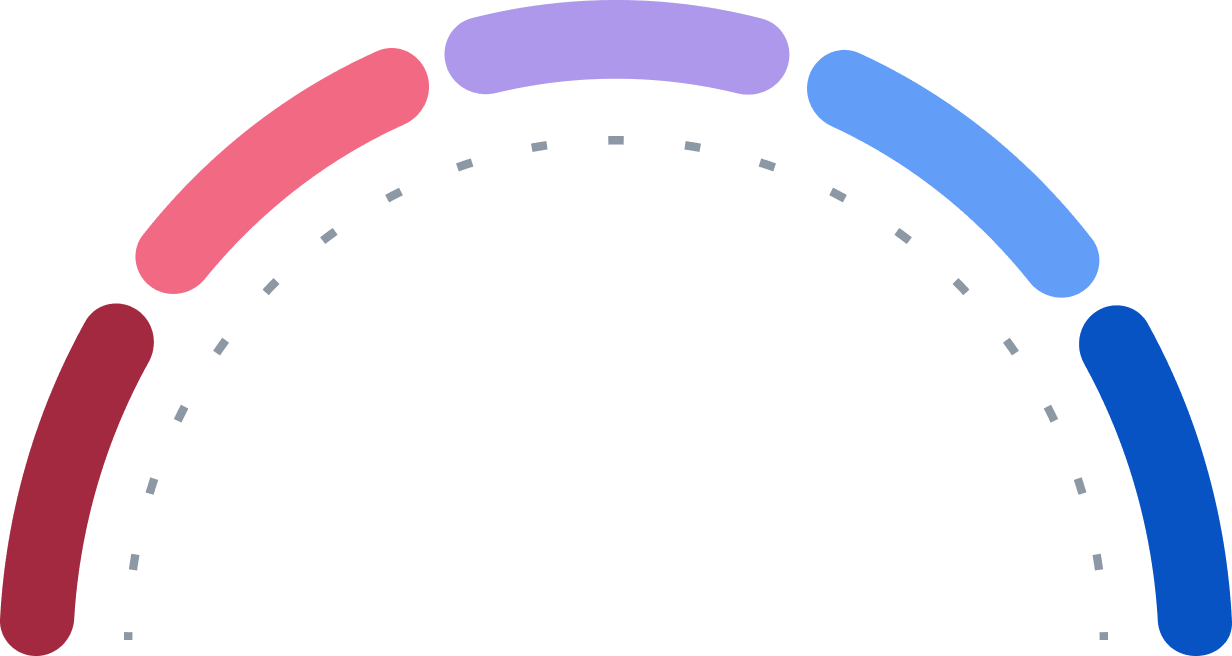
Buy
Strong Buy
Buy
Hold
Sell
Strong Sell
0
5
3
0
0


| Exchange | Ticker | Currency | Last Trade | Price | Daily Change |
|---|---|---|---|---|---|
NASDAQ |
SNGX
|
USD
|
01.05.2026 23:58
|
0,34 USD
| -0,005 USD
-1,45 %
|
IEX |
SNGX
|
USD
|
01.05.2026 19:58
|
0,35 USD
| 0,005 USD
+1,45 %
|
 Frankfurt
Frankfurt
| Date | Split |
|---|---|
| 06.06.2024 | 1:16 |
| 10.02.2023 | 1:15 |
| 30.12.2022 | 1.001:1 |
| 07.10.2016 | 1:10 |
| 01.02.2012 | 1:20 |
| 11.06.1997 | 1:15 |
| 06.12.1995 | 1:100 |
| Date | From | To |
|---|---|---|
| 30.09.2009 | DORB | SNGX |
| Name | Symbol |
|---|---|
| NASDAQ | SNGX |